Galvanizing is a method of applying a thin protective layer of zinc onto the surface of iron or steel components in order to prevent corrosion.
Hot-dip galvanizing is a specific galvanizing technique in which the zinc protective layer is applied by immersing the metallic component in a molten zinc bath. The zinc coating protects the steel product through barrier effect. The process includes several steps of preparation, cleaning, fluxing, hot-dip galvanizing, cooling, and quality inspection. Each step of the process is regulated in order to achieve optimum coating thickness, quality, appearance and performance based on ASTM International standard specifications.
The galvanized components are inexpensive alternatives to stainless steel, which are specifically designed for applications requiring high long-term corrosion resistance.
The higher electronegativity of zinc compared to iron enables it to continue with the protection of the product surface, even if the protective layer is somehow damaged. This is done via the preferential galvanic corrosion of zinc, also known as the galvanic effect. In this phenomenon, zinc can protect the adjacent steel by scarifying itself and this protection continues until the complete depletion of zinc.
Although zinc coating serves as a protective layer against the corrosion of the hot-dip galvanized steel, there is still a potential for galvanic corrosion through the formation of a bimetallic couple,once the hot-dip galvanized component comes into direct contact with other metallic structures. The formation of a bimetallic couple can destroy the galvanized steel by causing the rapid corrosion of zinc protective layer.
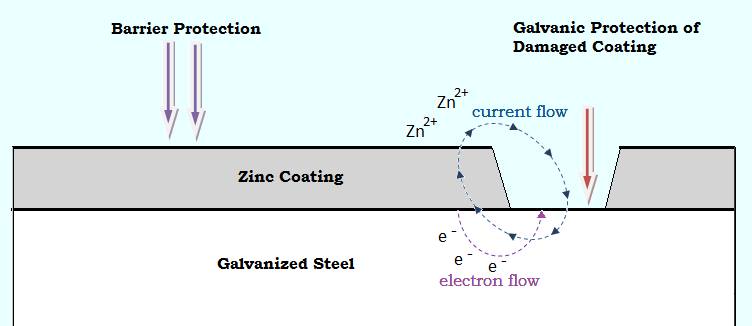
It is noteworthy that what defines a destructive corrosion is the depth of the corrosion penetration rather than the area affected by it. The severity of bimetallic corrosion highly depends on the type and position of the metal in contact with the hot-dip galvanized steel and to a lesser extent on the surface area of the two metals in contact. A large cathode surface in contact with a small anode surface can increase the penetration of corrosion in zinc. For instance, if a zinc rivet comes in contact with a steel plate, the galvanic corrosion of zinc would be severe due to the large surface area of steel (cathode) with respect to zinc (anode).
In another example, a direct contact between zinc and copper can result in the rapid corrosion of zinc due to the higher nobility of copper compared to zinc. If an electrical contact is formed between the two metals, the bimetallic corrosion would be inevitable. Even without an electrical contact, there is still a chance of corrosion if water flows from the noble metal towards zinc. In this case, the noble metal, such as copper, is partially dissolved in water and moves towards zinc with the water flow. If a contact between two such metals is necessary, then the formation of an electrical contact should be prevented by isolating connections and joints as well as making sure that the flow of water is directed from the galvanized component towards the noble metal and not vice versa.
The extent of corrosion also depends on the environment. For example, a hot-dip galvanized steel in contact with aluminium and its alloys suffers either no or insignificant corrosion in normal atmospheric conditions. However, if submerged in fresh water, moderate corrosion occurs and submersion in sea water should be avoided due to severe corrosion and damage of zinc and hot-dip galvanized steel.

Cadmium, magnesium and magnesium alloys are examples of suitable bimetallic couples with hot-dip galvanized steel, which can be used in air, fresh water, and sea water with no significant harm to the galvanized component or its coating.
